Last modified: 29 May, 2023
Introduction
Send this intervention to your inbox
This is a guide to point-of-care testing for hepatitis C (HCV) RNA. This guide provides an overview of how point-of-care HCV RNA testing works and how you can implement it in your service setting. We’ll give you some practical tools, tips, and resources and provide insights into the barriers you may face and how you might be able to overcome these barriers.
The implementation or scaling up of point-of-care HCV RNA testing is country and/or region-specific and the information we have provided is of a general nature.
The authors and reviewers of this guide include healthcare practitioners, researchers, laboratory scientists, policy makers and other service providers globally. This guide is part of our Intervention Toolkit which profiles innovative models of HCV care and offers guidance on how to set up different interventions for HCV in your service.
Definition
A point-of-care test is a test that is performed close to or near where a person is receiving care, has a fast time to result, and may lead to an immediate and informed change in patient management, potentially facilitating a reduced number of visits leading to increased linkage to care.
Point-of-care testing can enhance your model of care by reducing the barriers people may face to getting tested. Point-of-care tests can use multiple sample types including serum, plasma, venous whole-blood, capillary whole-blood and oral fluid, and can test for a variety of illnesses.
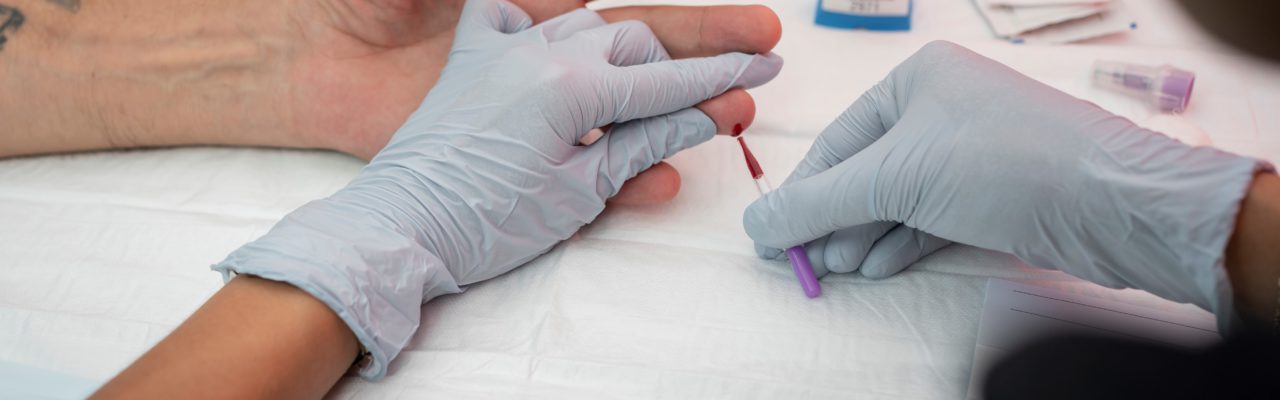
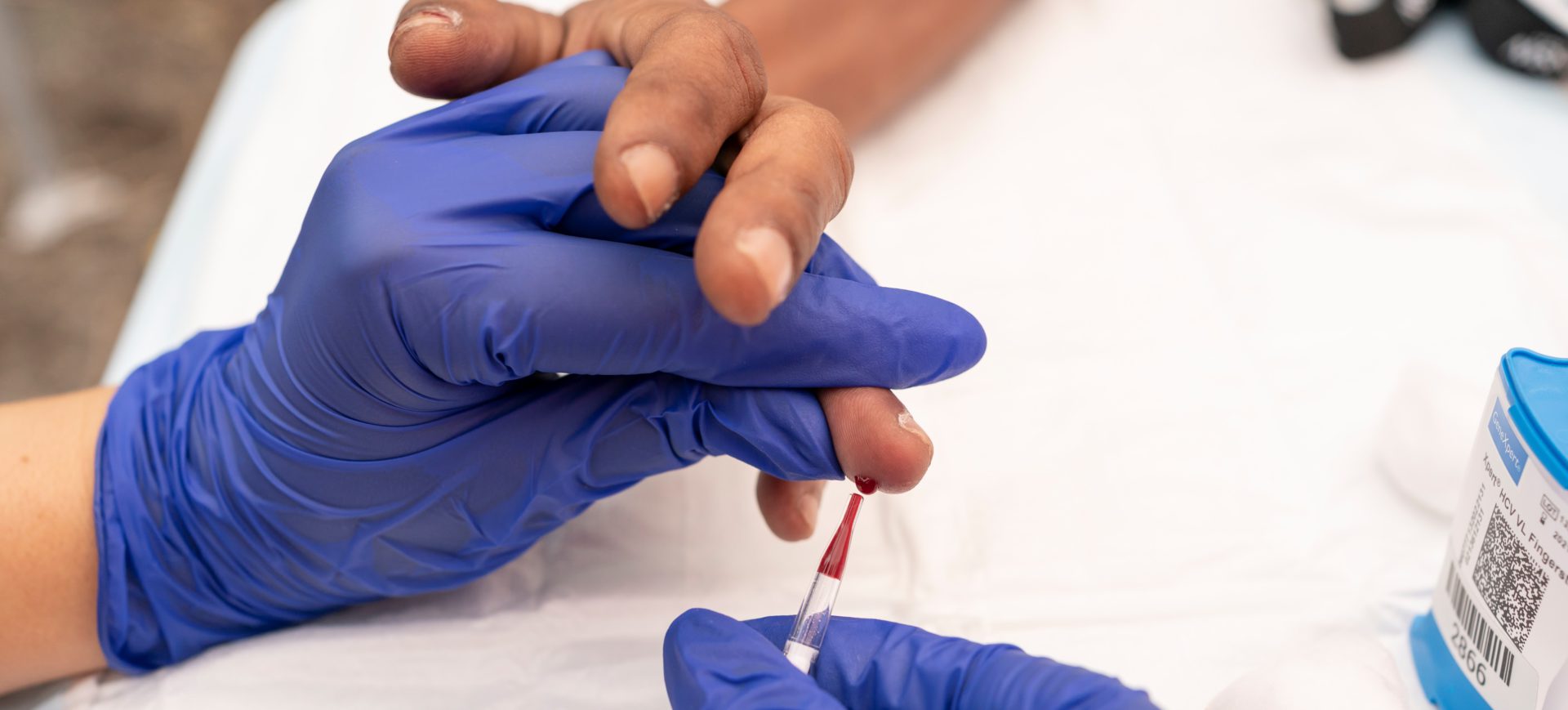
In this How-To Guide, we will focus on the Xpert® HCV Viral Load Fingerstick test, a point-of-care test which runs on the GeneXpert System, manufactured by Cepheid. This point-of-care test (or assay), tests for HCV RNA using finger-stick capillary whole blood.
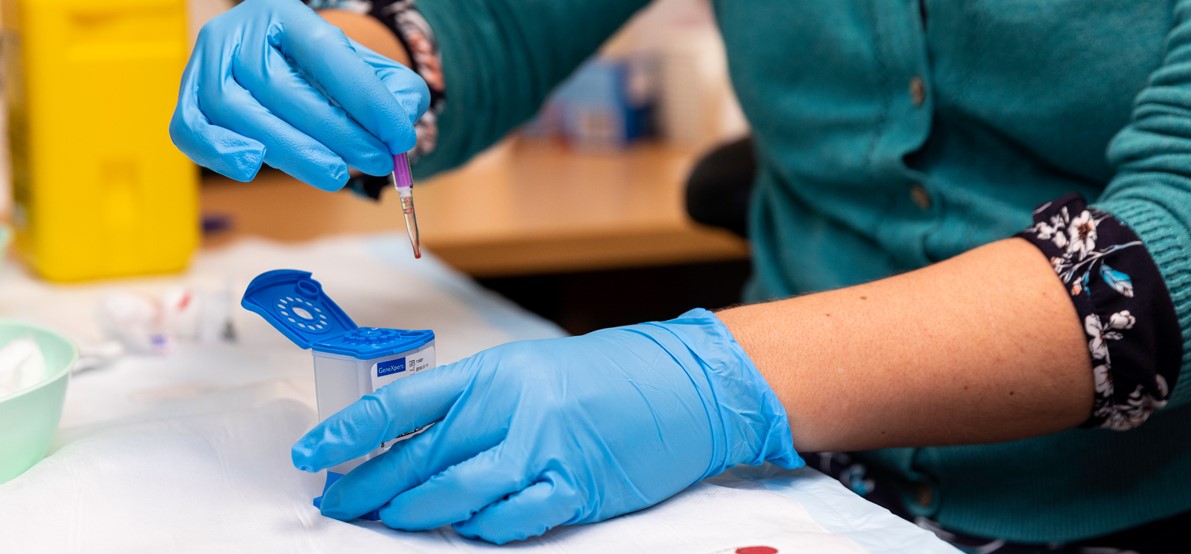
This type of testing for HCV involves collecting a capillary blood sample via a finger prick into a collection device (called a Minivette®), that is then dispensed into a cartridge and processed in a device called the ‘GeneXpert’, at or near the point-of-care testing site.
How does it work?
A blood sample for point-of-care HCV RNA testing can be collected and interpreted by trained health care professionals, frontline workers, and peers.
Blood is taken from a finger prick made with a lancet (a special medical blade with a sharp point) and capillary blood is collected into a Minivette®. The blood is dispensed into a cartridge, then loaded into the GeneXpert System, which generates an HCV RNA result within 60 minutes.
- Reduces the need for a venepuncture test. Venous access can be tricky, particularly in people with a history of injecting drug use, so finger prick testing can provide an alternate sample collection option.
- A smaller volume of blood is required than a venepuncture test.
- Has a fast time to result. The Xpert® HCV Viral Load Fingerstick test can deliver a result within 60 minutes.
- Reduces the number of times people need to see a healthcare professional. There are two tests required to confirm current HCV infection. First, an HCV antibody test tells us whether the person has ever been exposed to the virus. Second, an HCV RNA test confirms whether the person is currently infected with the virus. Point-of-care HCV RNA testing can provide a diagnosis of current infection from a single visit, thereby reducing the number of visits required.
- It can be done wherever the patient and equipment is, including prisons, needle and syringe programs, drug treatment settings, primary health care clinics, outreach settings and pharmacies, allowing people to be tested where they feel comfortable accessing care.
- Collection can be performed by peers or other people who are not practitioners, as long as they have received appropriate training and there is a plan in place to provide information and advice to inform the individual about the test results and linkage to care.
- It can be expensive. There are considerable costs with the procurement of a GeneXpert System, a laptop to interpret the results, ongoing costs of cartridges, annual maintenance contracts with the manufacturer, operator training, quality assurance programs, and IT systems to link the instrument to a network or laboratory information system.
- The GeneXpert will need to be maintained. The GeneXpert will require occasional service and maintenance visits. Software updates will be included in maintenance contracts however the availability of service engineers in a particular region may vary.
- There are likely to be stringent regulatory approvals and ongoing clinical governance is required.
- Appropriate training and quality management framework is required to ensure testing is accurate and safe. Proper use of the lancet and sample collection device ensures that an adequate sample is obtained without multiple punctures and minimise the number of invalid tests. Turnover of health service staff as well as limited capacity of health staff due to time restraints mean that ensuring adequate and sustainable training programs can be challenging.
How to set up point-of-care HCV RNA testing in your service
Availability: Check whether point-of-care HCV RNA tests (such as the Xpert HCV Viral Load Fingerstick test) are already being used in your region, and whether it is approved for use. In most places you can check this by contacting your local health department, or local pathology provider.
Policies and Guidelines: Consider national testing guidelines, reimbursement pathways, and private/public funding arrangements.
Clinical Governance: Clinical governance is the set of relationships and responsibilities established by a health service organisation between its local health department, governing body, executive, workforce, patients, consumers, and other stakeholders to ensure good clinical outcomes. Clinical governance includes patient safety, risk management and quality improvement. In some settings, clinical governance will be provided by the testing site, to ensure there is line management from the staff member undertaking testing- to the Clinic Director overseeing the facility. This is to ensure result delivery and follow-up is maintained for optimal client care. Consider contacting your local health department and pathology provider to find out who currently provides or could provide support and guidance for point-of-care testing.
Community Engagement: Include community such as local hepatitis organisations, local providers and other stakeholders in discussions that address whether the model of care you have selected in relation to point-of-care HCV RNA testing it is appropriate.
Procurement: Find out which point-of-care systems are being used in your region, level of vendor service support provided and if there is any financial support to purchase one. Financial partnerships with academic research departments or pharmaceutical companies may be available.
Site Selection criteria: Consider site suitability including power, space, temperature and infection control, where and how you will dispose of waste, as well as existing GeneXpert Systems.
Quality Management: Regulatory approval for use of point-of-care in your region may come with conditions attached including undertaking quality management processes. Development of Standard Operating Protocols ensures consistency, accuracy, and quality of testing. Staff training resources can be drawn from Standard Operating Protocols facilitating streamlining training. Monitoring of error and invalid rates may highlight areas for staff retraining. Find out what and how often quality control and external quality assurance needs to occur. If external quality assurance is a mandatory requirement for use, contact your laboratory who will be able to assist with enrolment. Quality control material (known positives and negatives) are best practice.
GeneXpert Systems and Xpert Assays: Identify GeneXpert System and Xpert assay supplier. Is the sales and support for the GeneXpert and Xpert HCV Viral Load Fingerstick tests provided directly by Cepheid in your country or is this managed by a local distributor? Will they install and maintain the instruments? Will they provide operator training, technical support, and remote monitoring? Will they provide support for quality assurance programs?
Clinical governance and quality assurance: For some point-of-care programs, clinical governance may be provided by jurisdictional Advisory Groups or Executive Committees, to offer ongoing guidance on clinical and cultural safety, analytical quality, data protection, privacy, training and programme integration . These committees may include representatives from Government, reference laboratories, peak bodies, and other programme staff. External quality assurance programs may be available from scientific organisations or professional colleges- contact your laboratory for further information or enrolment guidance.
Identify a laboratory that can offer support or reach out to your local health department for guidance.
Other suppliers: Identify other consumables you will need e.g. appropriate personal protective equipment (such as clean gloves and protective eye-wear at a minimum), disposable laboratory bench coat, lancets, Minivette® point-of-care test tubes, alcohol skin cleansing swabs, cotton wool balls, adhesive bandages, etc.
Key stakeholders: Identify who in your organization needs to be on board to develop a point-of-care HCV RNA testing program.
Evidence: Write a list of reasons why they should support this initiative – identify the benefits.
Partnerships: Consider partnerships with a range of stakeholders, would any of these stakeholders support your plan? In what way?
Elimination goals: Consider linking point-of-care HCV RNA testing to state, national or international strategic hepatitis elimination plans or goals
Risk management: Show management that you have thought about any possible risks. Identify any possible risks and demonstrate how you will manage them.
Budget: Put together a budget to show how much it will cost. Consider comparing the cost per point-of-care test and compare this to traditional testing methods.
Infrastructure: Identify what additional equipment you will need. Is there existing infrastructure you could use? Can you integrate this intervention into your existing services?
Staff, workflow and impact: Consider how you will staff the intervention. Can it work with your existing staff? Identify key staff who will be involved.
Training and protocols: Consider how you will train your staff, and who will run the training. Develop a relationship with a local laboratory or another quality assurance provider that can provide ongoing support for training.
Who will you test: Discuss with your team/manager who you will provide point-of-care testing for? Think about what other way you will test for blood borne virus infections such as hepatitis B and HIV. Are these assays approved in your country? Will you use point-of-care testing for this too? How will you assess liver fibrosis?
Helpful resources:
Record keeping: Decide how you will approach record keeping, so that you can easily identify who has been tested using a point-of-care test and how they have been linked to care if needed.
Connectivity and reporting systems: Consider data capture, notifications, electronic reporting, privacy, technical support, remote monitoring, tracking errors etc.
Client data storage: Consider where you will store client results and other required information for future reference – do you have client files already, or will you need a system to support this? Other required information may include assay lot number, date and time of test, system serial number and client information. Consider how the information will be securely stored and linked to a client’s medical record.
Policies and guidelines: Consider whether you will need to update any existing policies or guidelines within your service to include point-of-care testing.
Instruction manual: Consider where you will keep the instruction manual and maintenance manual for the point-of-care testing system. This will need to be kept updated, recording all ongoing maintenance, software updates and operational changes. For the GeneXpert System, the instruction manuals, maintenance schedule checklist, maintenance SOP, assay IFU’s etc will be stored on the laptop connected to the system. Cepheid (as manufacturer) will advise when software updates are required and will provide updated software packages to be installed with updated assay/kit versions.
Consent forms: Make sure you have appropriate consent forms that outline what the testing process is, what the results mean, and possible risks so that your client can make an informed decision.
Confidentiality: Consider how you will keep and maintain confidential records and ensure the privacy of your clients’ information.
Who will organise the supplies? Identify who will be responsible for the ordering of lancets, Minivette point-of-care test tubes, Xpert cartridges, and any other required supplies.
Who will maintain the equipment? Identify who will be responsible for ongoing maintenance of the GeneXpert System, downloading information and systems updates.
Who will perform the test? Identify who will perform the test – e.g. nurse, doctor, other health worker, peer, or various providers depending on the service setting and local regulations.
Who will provide the results? Identify who will provide results to your clients – are there any rules in your region about who is legally able to diagnose HCV?
How will you provide the results? Consider how you are going to provide test results to people, what support you can offer at that time and how you will link them into treatment if positive?
Who will notify positive results to the relevant body? If HCV is a notifiable disease in your region, identify who will be responsible for notifying the relevant body (e.g. your local public health department) of a positive result.
How will you link clients into treatment? Consider how you will link patients into treatment if they receive a positive result.
Training: You may need to consider training the relevant individuals on:
- How to perform the test
- How to interpret the results
- Infection control
- Client education and support
- Instrument maintenance and Connectivity solutions
Resources: Consider the tools that will support your staff and clients – these may be laminated infographics to show the client how the process works, client info sheets, and procedure manuals.
Helpful resources:
Who to test: Determine criteria for who you will test using point-of-care HCV RNA testing. Is point-of-care testing going to be used for testing all participants attending your service or just specific populations/settings? If point-of-care testing is only being used in specific populations, consider writing down an algorithm to define these situations. Develop a brief screening questionnaire to establish risk if you don’t already have this.
When to offer a test: Decide when to offer a point-of-care test to participants attending your service – is there a logical point in your interactions with people at which you can ask whether they would like to have a test?
Where to test: Decide how you will offer testing, where will the system be kept, when you will offer point-of-care testing, and who would be available to staff the testing program at these times.
At the testing site: Arrange a private area with cleanable surfaces, access to power source, where tests and consultation can be done, and the equipment can be securely and safely stored.
Consent: Make sure people having a point-of-care test will have access to the information and advice they will need. Ensure any instructions are provided in a way that makes it accessible for those who cannot read or have vision impairments.
Who will provide the results: Identify who will deliver results to your patients – are there any rules in your region about who is legally able to diagnose HCV and reporting to your public health unit? Are results delivered in person when the result is available (60 minutes)? The results need to be given by someone who is experienced and can provide information and support regarding positive HCV results and linkage to care.
Linkage to care: Identify the patient pathway to receiving care if they return a positive HCV RNA result. Consider whether that pathway will work for most of your clients, or will some require extra support?
Helpful resources:
Consider using an implementation theory, model, or framework: Using an implementation theory, model or framework can help guide the process of translating research into practice, understanding and/or explaining what influences implementation outcomes and evaluating implementation.
Order the supplies: Make a list of everything you will need to order.
Select implementation strategies: There are evidence-based strategies (methods, techniques) that you can use to promote and support the implementation and sustainability of point-of-care HCV RNA testing in your service. Here are a few examples:
- Build a coalition: Recruit and cultivate relationships with partners in the implementation effort. Are there existing local events or other services where you could create a partnership to offer a point-of-care testing service? For example, local shelters, drug treatment clinics, outreach services, etc.
- Identify and prepare champions: Identify and prepare individuals who dedicate themselves to supporting and marketing the implementation to overcome indifference or resistance. These champions can get the word out and increase awareness in your local population.
- Involve clients: Engage or include clients and their families in the implementation effort to highlight positive experience. Can you create a narrative of positive experiences from people who have taken the test?
- Involve and train peers: Engage or include trained peers in your model of care and in the implementation effort more broadly. Peers can provide support, promote point-of-care testing, collect point-of-care samples, and operate point-of-care testing systems if they are trained properly.
- Conduct educational meetings: Hold meetings with different stakeholder groups (e.g., providers, administrators, and community, client, and family stakeholders) to educate them about point-of-care testing.
- Conduct local consensus discussions: Include local providers and other stakeholders in discussions that address whether the model of care you have selected in relation to point-of-care testing is appropriate.
- Develop and disseminate educational materials: Develop and format toolkits and other supporting materials (in addition to the instruction manual) for stakeholders to learn about point-of-care testing and for clinicians to learn how to deliver it.
Helpful resources:
- INHSU – Peer Support Intervention
- INHSU – Patient Navigation Intervention (coming soon)
Evaluation plan: Plan how you will evaluate the uptake and effectiveness of the intervention. How will you know if the intervention has been useful? Consider developing a metric system generated periodically that allows you to track how many tests performed, positive results, treatment uptake and linkage to care.

Check with the manufacturer as first point of contact, to determine availability of the test. Your local public health department, local pathology provider, local medical microbiologist, and local hospital laboratory may also provide guidance, or try contacting other organisations in your country who have already explored point-of-care testing.
If they can’t point you in the right direction, contact us and we’ll do our best to help.
GeneXpert Systems may be being used for other disease testing. Find out if they could be integrated into HCV testing.
Approach testing manufactures to see if they would like to develop a service. Provide examples of other places who provide point-of-care testing. Consider private/public partnerships.
Consider other ways you might be able to access one. For example, by partnering with an academic institution as part of a research study, or by collaborating with another nearby service.
Point-of-care testing systems are used for many other diseases, not just HCV.
This is highly dependent on the health system you are in.
Check with your local public health department, local hospital laboratory, or try contacting other organisations in your country who have already explored point-of-care testing.
If they can’t point you in the right direction, contact us and we’ll do our best to help.
Contact other services who have successfully implemented point-of-care testing and ask them for advice. Use your networks to promote use, and ask peers, clients, and staff to promote it.
Rather than seek funding for a permanent program, consider approaching management for a pilot project with agreed upon endpoints and metrics that will help objectively show whether there is a benefit or not by using point-of-care testing.
Find examples of research that has demonstrated the benefits to clients.
Helpful resources:
Contact other services who have successfully implemented point-of-care testing and ask them for advice. You might not need funding – initiatives may be available or subsidised by governments or health departments.
If not, depending on the type of organization you are in, you may be able to seek grants to support implementation. Approaching pharmaceutical companies working in HCV may also be a possibility.
Helpful resources:
In some instances, instrument manufacturers may provide training of limited scope, though this may not be sufficient to realise maximum benefits of POC testing. Your local health department or local infectious disease clinicians may be able to provide training on the pre- and post-test counselling and education needed, including providing positive results and linkage to care.
Helpful resources:
Ensure training is appropriate and adequate. Run a few simulation clinic visits, where they can practice the process. Invite staff from places who are already using POC testing to provide advice and support and trouble shoot any issues that they identify.
Talk to the hepatology, gastroenterology, or infectious diseases department at your local hospital, they should be able to point you in the right direction. There may be community prescribers that would better suit your clients.
Discuss this with your local public health unit to get help regarding what other tests may be required and where the client can be referred if needed, to continue with care, treatment, and management of HCV if the test is positive.
Consider whether you can employ a peer to help get people on board and support clients through the testing process.
Videos
Resources
- Xpert® HCV VL Fingerstick Datasheet
- Simplification of testing and treatment in the quest to eliminate hepatitis C infection
- HCV GeneXpert Testing: Collection of Capillary Blood and Cartridge Preparation – National Australian Hepatitis C Point-of-Care Testing Program*
- Determine™ HBsAg 2 Testing: Collection of Capillary Blood and Patient Testing – National Australian Hepatitis C Point-of-Care Testing Program*
- Determine™ HIV Early Detect Testing: Collection of Capillary Blood and Patient Testing – National Australian Hepatitis C Point-of-Care Testing Program*
Disclaimers
*The following disclaimers refer to the resources listed above which are utilised as part of the National Australian Hepatitis C Point-of-Care Testing Program.
- POCT testing must only be performed by trained and competent operators.
- Research Use only refers to this assay not being certified by TGA as in vitro medical device.
- The GeneXpert finger stick HCV assay is a high risk (class IV) in vitro diagnostic test and must be used according to TGA specifications (e.g. trained operators and a quality testing framework).
Frequently asked questions 
What is a point-of-care test?
A point-of-care test is a test that is performed close to or near where a person is receiving care, has a fast time to result, and may lead to an immediate and informed change in patient management, potentially facilitating a reduced number of visits leading to increased linkage to care.
What is a GeneXpert System?
This is the system that runs the test and gives the result.
Does the client need to have venepuncture?
No, the test uses capillary blood from a finger prick.
How long does the test result take to be available?
The result is available 60 minutes after starting the instrument.
Do you need to be a doctor or nurse to perform the test?
No. This test can be performed by specifically trained healthcare workers and laboratory professionals
Got a question?
Contact us and we’ll do our best to help.
What does the evidence say?
Technical validation – sensitivity and specificity of point-of-care testing for HCV RNA:
Title |
Author |
Year |
Link |
| Diagnostic accuracy of assays using point-of-care testing or dried blood spot samples for the determination of HCV RNA: a systematic review | Catlett, Beth, et al. | 2022 | Read the paper |
| Diagnostic Accuracy of Point-of-Care HCV Viral Load Assays for HCV Diagnosis: A Systematic Review and Meta-Analysis | Tang, W, et al. | 2022 | Read the paper |
| Outcomes of the CT2 study: A ‘one-stop-shop’ for community-based hepatitis C testing and treatment in Yangon, Myanmar | Draper B, et al. | 2021 | Read the paper |
| Evaluation of the Xpert HCV VL Fingerstick point-of-care assay and dried blood spot HCV-RNA testing as simplified diagnostic strategies among people who inject drugs in Catalonia, Spain | Saludes, V, et al. | 2020 | Read the paper |
| Elbasvir and grazoprevir for hepatitis C virus genotype 1 infection in people with recent injecting drug use (DARLO-C): An open-label, single-arm, phase 4, multicentre trial | Grebely, J, et al. | 2020 | Read the paper |
| In-field evaluation of Xpert® HCV viral load Fingerstick assay in people who inject drugs in Tanzania | Mohamed, Zameer, et al. | 2019 | Read the paper |
| Evaluation of the Xpert HCV Viral Load Finger-Stick Point-of-Care Assay | Lamoury, F, et al. | 2018 | Read the paper |
| Evaluation of the Xpert HCV Viral Load point-of-care assay from venepuncture-collected and finger-stick capillary whole-blood samples: a cohort study | Grebely J, Lamoury FMJ, Hajarizadeh B, et al. | 2017 | Read the paper |
| Point -of -care testing (POCT) in molecular diagnostics: Performance evaluation of GeneXpert HCV RNA test in diagnosing and monitoring of HCV infection | Gupta E, Agarwala P, Kumar G, et al. | 2017 | Read the paper |
Impact of point-of-care RNA testing on HCV testing and linkage to treatment:
Title |
Author |
Year |
Link |
| Declining prevalence of current HCV infection and increased treatment uptake among people who inject drugs: The ETHOS Engage study | Valerio, Heather, et al | 2022 | Read the paper |
| Feasibility and effectiveness of HCV viraemia testing at harm reduction sites in Georgia: A prospective three-arm study | Shilton, Sonjelle | 2022 | Read the paper |
| “That was quick, simple, and easy”: Patient perceptions of acceptability of point-of-care hepatitis C RNA testing at a reception prison | Lafferty, Lise, et al | 2022 | Read the paper |
| Outcomes of the CT2 study: A ‘one-stop-shop’ for community-based hepatitis C testing and treatment in Yangon, Myanmar | Draper, Bridget Louise, et al. | 2021 | Read the paper |
| Detection of active hepatitis C in a single visit and linkage to care among marginalized people using a mobile unit in Madrid, Spain | Ryan, Pablo, et al. | 2021 | Read the paper |
| A decentralised point-of-care testing model to address inequities in the COVID-19 response | Hengel, Belinda, et al. | 2020 | Read the paper |
| Time matters: Point of care screening and streamlined linkage to care dramatically improves hepatitis C treatment uptake in prisoners in England | Mohamed, Zameer, et al. | 2020 | Read the paper |
| Novel Hepatitis C Virus (HCV) Diagnosis and Treatment Delivery Systems: Facilitating HCV Elimination by Thinking Outside the Clinic | Bajis, S, et al. | 2020 | Read the paper |
| Rapid test & treat programme successfully facilitating hepatitis C micro-elimination in a women’s prison | Ustianowski A, et al. | 2020 | Read the paper |
| Approaches to providing hepatitis C viremia testing to people who inject drugs in Georgia, HEAD START (Hepatitis Elimination Through Access to Diagnostics) Georgia | Japaridze M, et al. | 2019 | Read the paper |
| Acceptability and preferences of point-of-care finger-stick whole-blood and venepuncture hepatitis C virus testing among people who inject drugs in Australia | Bajis, Sahar, et al. | 2018 | Read the paper |
| Hepatitis C point-of-care diagnostics: in search of a single visit diagnosis | Grebely J, et al. | 2017 | Read the paper |
Implementation theories:
Title |
Author |
Year |
Link |
| Making sense of implementation theories, models and frameworks | Nilsen P. | 2015 | Read the paper |
| A refined compilation of implementation strategies: results from the Expert Recommendations for Implementing Change (ERIC) project | Powell B, et al. | 2015 | Read the paper |
| Effective Practice and Organization of Care (EPOC) | Shepperd S, et al. | 2015 | Read the paper |
Photos used with permission from the Kirby Institute, UNSW, Sydney, Australia